Entry
Reader's guide
Entries A-Z
Subject index
In Vitro Fertilization
IN VITRO FERTILIZATION (IVF) involves the combination of an unfertilized oocyte and spermatozoa in a Petri dish to allow fertilization to occur. Subsequent cleavage of the newly formed zygote can be supported in vitro by various culture conditions. Transfer of embryos into the uterus has resulted in pregnancy and live births in many mammalian species. Embryos cultured to the blastocyst stage have provided a source for embryonic stem cells. Embryonic stem cells have the capacity to renew and to differentiate into many cells of the body, a term known as potency. Therefore, in vitro fertilization provides a clinical application for infertility issues as well as a research avenue to understand early development.
Although the process of reproduction has been of interest to both science and philosophy since Aristotle's time, the first example of in vitro fertilization was shown by Lazaro Spallanzani (1729–99) when he demonstrated that tadpoles only developed if oocytes came into contact with semen. Much later, Yanagimachi and Chang (1963) reported the fertilization of hamster oocytes with capacitated sperm which prefaced the successes in human in vitro fertilization. The first live birth from human in vitro fertilization was in 1978, by Patrick Steptoe and Robert Edwards in the United Kingdom. Since then, the in vitro fertilization procedure has become a routine treatment option for patients dealing with infertility.
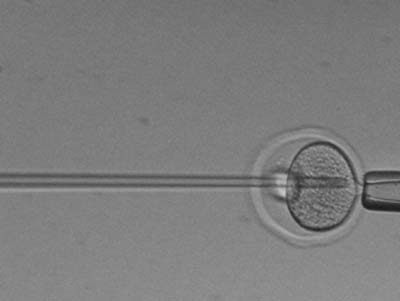
In vitro embryo culture has helped researchers identify markers associated with pluripotency such as Oct-4.
Procedure
The first step in the in vitro fertilization process involves the superovulation of the female. In current paradigms, this entails the down—regulation of gonadotrophic—releasing hormone from the hypothalamus which suppresses the anterior pituitary from secreting the gonadotrophins: follicle stimulating hormone and luteinizing hormone. This results in a temporary state of hypogonadotrophic hypogonadism. Once this state is reached, gonad—otrophin stimulation can begin by the administration of follicle stimulating hormone to induce follicle growth in the ovaries. During this time, follicle growth is monitored via ultrasound and by peripheral blood levels of estradiol.
Once follicle size and estradiol levels have reached the desirable size and levels, respectively, human chorionic gonadotrophin is given for ovulation induction. Ovulation in normal cycles is preceded by a surge in luteinizing hormone. Since the β-subunit of human chorionic gonadotrophin is similar to that of luteinizing hormone, human chorionic gonadotrophin is often used in lieu of luteinizing hormone. Once ovulation has been induced, oocyte collection is scheduled to occur within 36 hours postinjection. This 36-hour window is critical because during this time, the oocytes within the follicles will reinitiate meiosis II and will progress to metaphase II.
During oocyte collection, semen will be obtained by masturbation and will be processed typically by gradient centrifugation to remove the seminal plasma and debris. Next, the oocytes and sperm will be combined in a Petri dish to undergo fertilization. An alternative approach is to perform intracytoplasmic sperm injection where an isolated sperm cell is injected into each oocyte. This helps the sperm bypass the zona pellucida of the oocyte and results in high rates of fertilization. Successful fertilization is evidenced by the appearance of two pronuclei in the oocyte at approximately 16–18 hours postinsemination. The pronuclei are said to come into syngamy where the nucleus from the oocyte comes into close proximity with the nucleus from the spermatocyte.
...
- Biology
- Biotechnology, History of
- Cell Sorting
- Cells, Adult
- Cells, Amniotic
- Cells, Developing
- Cells, Embryonic
- Cells, Fetal
- Cells, Human
- Cells, Monkey
- Cells, Mouse (Embryonic)
- Cells, Neural
- Cells, Sources of
- Cells, Umbilical
- Cytogenetic Instability of Stem Cells
- Developmental Biology
- Differentiation, In Vitro and In Vivo
- Division Types (Symmetrical and Asymmetrical)
- Experimental Models
- Feeder/Feeder—Free Culture
- Gut Stem Cells
- Induced Pluripotent Stem Cells
- Lineages
- Mammary Stem Cells
- Markers of Sternness
- Methods of Growing Cells
- Microenvironment and Immune Issues
- Neuralstem
- Neurosphere Cultures
- Niche Self—Renewal
- Nuclear Reprogramming
- Parthogenesis
- Plant Stem Cells
- Prostate Tissue Stem Cells
- Renal Stem Cells
- Self—Renewal, Stem Cell
- Stem Cell Applications, Articular Cartilage
- Stem Cell Applications, Tendon and Ligament
- Stem—Like Cells, Human Brain
- Tissue Culture
- Transdifferentiation
- Clinical Trials
- Clinical Trials Outside U.S.: Amyotrophic Lateral Sclerosis
- Clinical Trials Outside U.S.: Avascular Necrosis
- Clinical Trials Outside U.S.: Severe Coronary Artery Disease
- Clinical Trials Outside U.S.: Spinal Cord Injury
- Clinical Trials Within U.S.: Batten Disease
- Clinical Trials Within U.S.: Blind Process
- Clinical Trials Within U.S.: Cancer
- Clinical Trials Within U.S.: Heart Disease
- Clinical Trials Within U.S.: Peripheral Vascular Disease
- Clinical Trials Within U.S.: Skin Transplants (Burns)
- Clinical Trials Within U.S.: Spinal Cord Injury
- Clinical Trials Within U.S.: Traumatic Brain Injury
- Clinical Trials Worldwide
- Countries
- Diseases
- Ethics
- History and Technology
- Birth Dating of Cells by Retrovirus
- Bone Marrow Transplants
- BrdU/Thymidine
- Fluorescence—Activated Cell Sorting
- Human Embryonic Stem Cells
- In Vitro Fertilization
- Mouse ES Cell Isolation
- MRI Tracking
- Non—Human Primate Embryonic Stem Cells
- Nuclear Transfer, Altered
- Nuclear Transfer, Somatic
- Parthogenesis
- Preimplantation Genetic Diagnosis
- Viral Vectors: Adeno—Associated Virus
- Viral Vectors: Adenovirus
- Viral Vectors: Lentivirus
- Industry
- Institutions
- Albert Einstein College of Medicine
- Baylor College of Medicine
- Bonn University
- Burnham Institute
- Caltech
- Cambridge University
- Case Western Reserve University/Cleveland Clinic
- Children's Hospital, Boston
- Columbia University
- Coriell Institute
- Duke University
- Genetics Policy Institute
- Harvard University
- Indiana University
- Johns Hopkins University
- Kyoto University
- Massachusetts General Hospital
- Massachusetts Institute of Technology
- Mayo Clinic
- McMaster University
- Mount Sinai School of Medicine
- National Academy of Science
- Northwestern University
- Oregon Health & Science University
- Ottawa Health Research Institute
- Oxford University
- Princeton University
- Reeve—Irvine Research Center
- Robarts Research Institute
- Rockefeller University
- Rutgers University
- Salk Institute
- Scripps Research Institute
- Sloan—Kettering Institute
- Stanford University
- Stowers Institute
- University of California, Berkeley
- University of California, Davis
- University of California, Los Angeles
- University of California, San Diego
- University of California, San Francisco
- University of Connecticut
- University of Georgia
- University of Miami
- University of Michigan
- University of Minnesota
- University of North Carolina, Chapel Hill
- University of Pittsburgh
- University of Southern California
- University of Texas Health Science Center at Houston
- University of Toronto
- University of Washington/Hutchinson Cancer Center
- University of Wisconsin, Madison
- Vanderbilt of University
- Wake Forest University
- Weill—Cornell Medical College
- Whitehead Institute
- Yale University
- Legal Issues
- Organizations
- American Association for the Advancement of Science
- Australian Stem Cell Centre
- California Institute for Regenerative Medicine
- Canadian Stem Cell Network
- China Stem Cell News
- Christopher Reeve Foundation
- Community of Stem Cell Scientists
- Danish Stem Cell Research Center
- East of England Stem Cell Network
- European Consortium for Stem Cell Research—EuroStemCell
- International Society for Stem Cell Research
- International Stem Cell Forum
- Japan Human Cell Society
- Lasker Foundation
- Medical Research Council UK Stem Cell Initiative
- Michael J. Fox Foundation
- National Institutes of Health
- National Stem Cell Bank
- Parkinson's Disease Foundation
- Scottish Stem Cell Network
- Stem Cell Genome Anatomy Projects
- Swiss Stem Cell Network
- UK National Stem Cell Network
- Wisconsin Alumni Research Foundation
- People
- Alvarez—Buylla, Arturo
- Anversa, Piero
- Charo, Robin Alta
- Eaves, Connie
- Eggan, Kevin
- Fuchs, Elaine
- Gage, Fred
- Gearhart, John
- Goldman, Steven A.
- Jaenisch, Rudolf
- Keller, Gordon
- Kriegstein, Arnold
- Lanza, Robert
- Losordo, Douglas
- Macklis, Jeffrey
- McKay, Ronald D. G.
- Melton, Doug
- Morrison, Sean
- Mummery, Christine
- Nottebohm, Fernando
- Okano, Hideyuki
- Orkin, Stuart
- Rao, Mahendra
- Smith, Austin
- Snyder, Evan
- Steindler, Dennis A.
- Studer, Lorenz P.
- Thomson, James
- Van der Kooy, Derek
- Verfaillie, Catherine
- Vescovi, Angelo
- Weissman, Irving
- Wilmut, Ian
- Politics
- Advocacy
- Coalition for the Advancement of Medical Research
- Congress: Votes and Amendments (Cloning/Embryos)
- Dickey Amendment
- Do No Harm: The Coalition of Americans for Research Ethics
- National Right to Life Committee
- President's Council on Bioethics
- Presidential Campaigns
- Reagan, Nancy
- Special Interest/Lobby Groups
- Stem Cells, Bush Ruling
- Religion
- States
- Alabama
- Arizona
- Arkansas
- California
- Colorado
- Connecticut
- Delaware
- Florida
- Georgia
- Hawaii
- Idaho
- Illinois
- Indiana
- Iowa
- Kansas
- Kentucky
- Louisiana
- Maine
- Maryland
- Massachusetts
- Michigan
- Minnesota
- Mississippi
- Missouri
- Montana
- Nebraska
- Nevada
- New Hampshire
- New Jersey
- New Mexico
- New York
- North Carolina
- North Dakota
- Ohio
- Oklahoma
- Oregon
- Pennsylvania
- Rhode Island
- South Carolina
- South Dakota
- Tennessee
- Texas
- Utah
- Vermont
- Virginia
- Washington
- West Virginia
- Wisconsin
- Wyoming
- Loading...
Get a 30 day FREE TRIAL
-
Watch videos from a variety of sources bringing classroom topics to life
-
Read modern, diverse business cases
-
Explore hundreds of books and reference titles

Sage Recommends
We found other relevant content for you on other Sage platforms.

Have you created a personal profile? Login or create a profile so that you can save clips, playlists and searches