Entry
Reader's guide
Entries A-Z
Subject index
Salinity
TWO ATTRIBUTES OF the oceans, temperature and salinity, determine the density of seawater, and the differences in density between the water masses in the worlds oceans causes the water to flow in ther-mohaline circulation, thereby producing the greatest oceanic current on the planet.
Salinity is the distinct taste of seawater and is the result of the presence of dissolved salts (more than 85 dissolved constituents), among which chloride (Cl) and sodium (Na), the elements of common table salt, are the most abundant. The term salinity refers to the content of these dissolved salts and has been defined as grams of dissolved salts per kilogram of seawater. Salinity has been expressed as parts per thousand (‰ or ppt) and, more recently, by practical salinity units (psu).
On average, a kilogram of seawater has 35 grams of dissolved salts, so its salinity content is 35‰, or 35 psu. The accuracy of most laboratory salinometers (see below) is about 0.001 psu. Thus, only those components with a concentration over 0.001‰ will contribute to such salinity estimates. Only 15 of the dissolved salts have concentrations above that limit.
A key observational result (known as the principle of Dittmar, after William Dittmar, a Scottish professor of chemistry; the principle of Maury, after Matthew Fontaine Maury, an American astronomer, oceanographer, and geologist; or the hypothesis of Forchhammer, after Johan Georg Forchhammer, a Danish mineralogist and geologist) is that the relative concentration between some of these most abundant salts is virtually constant over much of the World Ocean. This finding indicates that the physical characterization of seawater is given by its temperature, pressure, and a single number reflecting the concentration of the most abundant components. Salinity is that number.
Measuring Salinity
In 1902, an international commission defined salinity as the total amount of solid material, in grams, contained in 1 kg. of seawater when all the carbonate has been converted to oxide, the bromide and iodine replaced by chlorine, and all organic matter completely oxidized. With this definition in hand, the commission estimated the salinity of several seawater samples and the fixed relationship between salinity (5) and chlorinity:
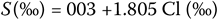
This was known as Knudsen's equation, after Martin Hans Christian Knudsen, a Danish physicist (1871–1949), and was redefined in 1969 as
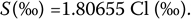
Defining salinity in terms of chlorinity alleviates the practical difficulties of measuring salinity through evaporating water samples to dryness. For calibration purposes, artificial water with salinity almost equal to 35‰, known as Copenhagen water, is manufactured to serve as a reference. Copenhagen water has a chlorinity of 19.381‰. This approach requires the chemical titration of water samples usually obtained by Nansen bottles, named after Fridtjof Bedel-Jarlsberg Nansen, a Norwegian explorer and scientist (1861–1930), which are self-closing containers that collect water from different depths.
Pure water is a poor electrical conductor. However, the presence of dissolved salts greatly increases its conductivity, which, in fact, is a function of pressure, temperature, and the degree of ionization of the dissolved salts. In the second half of the 20th century, technical improvements in the measurement of the electrical conductivity of seawater led to the development of the so-called salinometers. Conductive sali-nometers measure the ratio between the conductivity of the sample against that of a reference sample of known salinity. Researchers using conductivity salinometers in the beginning were giving a salinity value of 35‰ to any sample having the same conductivity as the Copenhagen water, even though the mass of salt per kilogram of water was not guaranteed to be the same in both cases. This was because conductivity depends on the degree of ionization of the dissolved salts and not on the absolute mass of salt. In 1978, the salinity scale was redefined in terms of the conductivity ratio, K15, between any given sample and the reference
...
- Atmospheric Sciences
- Aerosols
- Anticyclones
- Atmospheric Absorption of Solar Radiation
- Atmospheric Boundary Layer
- Atmospheric Composition
- Atmospheric Emission of Infrared Radiation
- Atmospheric General Circulation Models
- Clouds, Cirrus
- Clouds, Cumulus
- Clouds, Stratus
- Condensation
- Convection
- Coriolis Force
- Cyclones
- Doldrums
- Evaporation and Transpiration
- Evolution of the Atmosphere
- Hadley Circulation
- Heat, Latent
- Heat, Sensible
- Hurricanes and Typhoons
- Hydrological Cycle
- Intertropical Convergence Zone
- Jet Streams
- Mesosphere
- Monsoons
- Precipitation
- Radiation, Absorption
- Radiation, Infrared
- Radiation, Long Wave
- Radiation, Microwave
- Radiation, Short Wave
- Radiation, Ultraviolet
- Rain
- Stratosphere
- Thermosphere
- Thunderstorms
- Trade Winds
- Troposphere
- Walker Circulation
- Waves, Gravity
- Waves, Internal
- Waves, Kelvin
- Waves, Planetary
- Waves, Rossby
- Weather
- Winds, Easterlies
- Winds, Westerlies
- Climate
- Abrupt Climate Changes
- Aerosols
- Albedo
- Anthropogenic Forcing
- Carbon Cycle
- Chaos Theory
- Climate Cycles
- Climate Forcing
- Climate Thresholds
- Climate Zones
- Climatic Data, Atmospheric Observations
- Climatic Data, Cave Records
- Climatic Data, Historical Records
- Climatic Data, Ice Observations
- Climatic Data, Instrumental Records
- Climatic Data, Lake Records
- Climatic Data, Nature of the Data
- Climatic Data, Oceanic Observations
- Climatic Data, Proxy Records
- Climatic Data, Sea Floor Records
- Climatic Data, Sediment Records
- Climatic Data, Tree Ring Records
- Detection of Climate Changes
- Earthshine
- El Niño and La Niña
- Gaia Hypothesis
- Global Warming
- Greenhouse Effect
- Greenhouse Gases
- History of Climatology
- History of Meteorology
- Hydrological Cycle
- Internal Climate Variability
- Methane Cycle
- Milankovitch Cycles
- Monsoons
- North Atlantic Oscillation
- Oxygen Cycle
- Seasonal Cycle
- Southern Oscillation
- Sunlight
- Thermodynamics
- Volcanism
- World Systems Theory
- Climate and Society
- An Inconvenient Truth
- Adaptation
- Alternative Energy, Ethanol
- Alternative Energy, Solar
- Alternative Energy, Wind
- Automobiles
- Bush (George W.) Administration
- Capitalism
- Clean Air Act, U.S.
- Clinton Administration
- Coal
- Conservation
- Culture
- Ecological Footprint
- Education
- Environmental Protection Agency (EPA)
- Framework Convention on Climate Change
- Geography
- Globalization
- Greenhouse Effect
- Greenhouse Gases
- Gross National Product
- Health
- Industrialization
- Maximum Sustainable Yield
- Measurement and Assessment
- Media, Books and Journals
- Media, Internet
- Media, TV
- Movements, Environmental
- Needs and Wants
- Nuclear Power
- Oil, Consumption of
- Policy, U.S.
- Population
- Preparedness
- Public Awareness
- Regulation
- Religion
- Resources
- Risk
- Social Ecology
- Sustainability
- Technology
- Tourism
- Climate Change, Effects
- Adaptation
- Agriculture
- Animals
- Arctic Ocean
- Attribution of Global Warming
- Aviation
- Desertification
- Deserts
- Diseases
- Drought
- Economics, Cost of Affecting Climate Change
- Economics, Impact From Climate Change
- Ecosystems
- Floods
- Food Production
- Glaciers, Retreating
- Hurricanes and Typhoons
- Impacts of Global Warming
- Oceanic Changes
- Plants
- Polar Bears
- Rainfall Patterns
- Sea Level, Rising
- Species Extinction
- Thermohaline Circulation
- Transportation
- Tsunamis
- Climate Feedbacks
- Climate Models
- Atmospheric Component of Models
- Climate Model
- Computer Models
- Energy Balance Models
- Historical Development of Climate Models
- Ice Component of Models
- Land Component of Models
- Modeling of Ice Ages
- Modeling of Ocean Circulation
- Modeling of Paleoclimates
- Ocean Component of Models
- Simulation and Predictability of Seasonal and Interannual Variations
- Validation of Climate Models
- Countries: Africa
- Algeria
- Angola
- Benin
- Botswana
- Brunei Darussalam
- Burkina Faso
- Burundi
- Côte d'Ivoire
- Cameroon
- Cape Verde
- Central African Republic
- Chad
- Comoros
- Congo
- Congo, Democratic Republic of
- Djibouti
- Egypt
- Equatorial Guinea
- Eritrea
- Ethiopia
- Gabon
- Gambia
- Ghana
- Guinea
- Guinea-Bissau
- Kenya
- Lesotho
- Liberia
- Madagascar
- Malawi
- Mali
- Mauritania
- Mauritius
- Morocco
- Mozambique
- Namibia
- Niger
- Nigeria
- Rwanda
- São Tomé and Principe
- Senegal
- Sierra Leone
- Somalia
- South Africa
- Sudan
- Swaziland
- Tanzania
- Togo
- Tunisia
- Uganda
- Zambia
- Zimbabwe
- Countries: Americas
- Antigua and Barbuda
- Argentina
- Bahamas
- Barbados
- Belize
- Bolivia
- Brazil
- Canada
- Chile
- Colombia
- Costa Rica
- Cuba
- Dominica
- Dominican Republic
- Ecuador
- El Salvador
- Grenada
- Guatemala
- Guyana
- Haiti
- Honduras
- Jamaica
- Mexico
- Nicaragua
- Panama
- Paraguay
- Peru
- Saint Kitts and Nevis
- Saint Lucia
- Saint Vincent and the Grenadines
- Suriname
- Trinidad and Tobago
- United States of America
- Uruguay
- Venezuela
- Countries: Asia
- Afghanistan
- Azerbaijan
- Bahrain
- Bangladesh
- Bhutan
- Cambodia
- China
- East Timor
- Georgia (Nation)
- India
- Indonesia
- Iran
- Iraq
- Israel
- Japan
- Jordan
- Kazakhstan
- Korea, North
- Korea, South
- Kuwait
- Kyrgyzstan
- Laos
- Lebanon
- Malaysia
- Maldives
- Mongolia
- Myanmar
- Nepal
- Oman
- Pakistan
- Philippines
- Qatar
- Russia
- Saudi Arabia
- Seychelles
- Singapore
- Sri Lanka
- Syria
- Tajikistan
- Thailand
- Turkey
- Turkmenistan
- Ukraine
- United Arab Emirates
- Uzbekistan
- Vietnam
- Yemen
- Countries: Europe
- Albania
- Andorra
- Armenia
- Austria
- Belarus
- Belgium
- Bosnia and Herzegovina
- Bulgaria
- Croatia
- Cyprus
- Czech Republic
- Denmark
- Estonia
- Finland
- France
- Germany
- Greece
- Hungary
- Iceland
- Ireland
- Italy
- Latvia
- Liechtenstein
- Lithuania
- Luxembourg
- Macedonia (FYROM)
- Malta
- Moldova
- Monaco
- Netherlands
- Norway
- Poland
- Portugal
- Romania
- San Marino
- Serbia and Montenegro
- Slovakia
- Slovenia
- Spain
- Sweden
- Switzerland
- United Kingdom
- Countries: Pacific
- Glaciology
- Government and International Agencies
- Air Force, U.S.
- Department of Defense, U.S.
- Department of Energy, U.S.
- Department of State, U.S.
- Geophysical Fluid Dynamics Laboratory
- Global Atmospheric Research Program (GARP)
- Goddard Institute for Space Studies
- National Aeronautics and Space Administration (NASA)
- National Oceanic and Atmospheric Administration (NOAA)
- National Science Foundation
- Navy, U.S.
- Office of Naval Research
- United Nations
- World Health Organization
- World Meteorological Organization
- Institutions Studying Climate Change
- Alaska Climate Research Center
- Alliance to Save Energy
- American Council for an Energy-Efficient Economy
- American Electric Power
- American Gas Association
- American Geophysical Union
- American Meteorological Society
- Antarctic Meteorology Research Center
- Applied Energy Services, Inc.
- Atmosphere, Climate and Environment Information Programme (UK)
- Atmospheric Research and Information Centre
- BP
- Canadian Association for Renewable Energies
- Cantor Fitzgerald EBS
- Center for Clean Air Policy
- Center for Energy Efficiency
- Center for International Climate and Environmental Research
- Center for International Environmental Law
- Center for Ocean-Atmospheric Prediction Studies
- Center for Science and Environment (India)
- Climate Action Network
- Climate Change Knowledge Network
- Climate Impacts LINK Project
- Climatic Research Unit
- Colorado Climate Center
- Cooperative Institute for Arctic Research
- Cornell University
- David Suzuki Foundation
- Department of Energy, U.S.
- Desert Research Institute
- Edison Electric Institute
- Environmental and Societal Impacts Group
- Environmental Defense
- Environmental Development Action in the Third World
- Environmental Financial Products, LLC
- Environmental Protection Agency (EPA)
- European Commission
- FEEM (Italy)
- Florida State University
- Foundation for International Environmental Law and Development
- Friends of the Earth
- Geophysical Fluid Dynamics Laboratory
- Global Atmospheric Research Program (GARP)
- Global Environment Facility (GEF)
- Global Industrial and Social Progress Research Institute (GISPRI)
- Greenpeace International
- Harvard University
- Heinz Center
- Idaho State Climate Services
- Indiana University
- Institute of Energy Economics (Argentina)
- Intergovernmental Panel on Climate Change (IPCC)
- International Council of Scientific Unions (ICSU)
- International Energy Agency (IEA)
- International Institute for Sustainable Development (IISD)
- International Research Institute for Climate Prediction
- International Solar Energy Society (ISES)
- International Union of Geodesy and Geophysics (IUGG)
- Joint Institute for the Study of the Atmosphere and Ocean (JISAO)
- Kyoto Mechanisms
- LDEO Climate Modeling Group
- Marshall Institute
- Midwestern Regional Climate Center
- National Academy of Sciences, U.S.
- National Association of Energy Service Companies (NAESCO)
- National Center for Atmospheric Research (NCAR)
- Natsource
- Natural Resources Defense Council (NRDC)
- New Mexico Climate Center
- OECD Annex 1 Expert Group on the UNFCCC
- OECD Climate Change Documents
- Ohio State University
- Oregon Climate Service
- Oregon State University
- Organisation for Economic Co-operation and Development (OECD)
- Penn State University
- Pew Center on Global Climate Change
- Renewable Energy Policy Project (REPP)
- Resources for the Future (RFF)
- Royal Dutch/Shell Group
- Royal Meteorological Society
- Scripps Institute of Oceanography
- Solar Energy Industries Association (SEIA)
- Stockholm Environment Institute (SEI)
- Tata Energy Research Institute (TERI)
- Trexler and Associates, Inc.
- UN Conference on Trade and Development/Earth Council Institute: Carbon Market Program
- United Nations Development Programme (UNDP)
- United Nations Environment Programme (UNEP)
- University Corporation for Atmospheric Research
- University Corporation for Atmospheric Research Joint Office for Science Support
- University of Arizona
- University of Birmingham, Meteorology and Climatology Department
- University of California
- University of Colorado
- University of Delaware, Center for Climatic Research
- University of Florida
- University of Hawaii, School of Ocean and Earth Science and Technology
- University of Illinois, Department of Atmospheric Sciences
- University of Kentucky, Agricultural Weather Center
- University of Leeds, Institute for Atmospheric Science
- University of Maine, Institute for Quaternary Studies
- University of Maryland, Department of Meteorology
- University of Miami
- University of Michigan
- University of New Hampshire
- University of Oklahoma, Weather Radar
- University of Reading, Department of Meteorology
- University of Utah, Department of Meteorology
- University of Washington, Atmospheric Science Department
- Utah Climate Center
- Weather World 2010 Project
- Western Regional Climate Center
- Woods Hole Oceanographie Institute
- World Bank
- World Business Council for Sustainable Development
- World Meteorological Organization
- World Resources Institute
- World Wildlife Fund
- Worldwatch Institute
- Oceanography
- Agulhas Current
- Antarctic Circumpolar Current
- Arctic Ocean
- Atlantic Ocean
- Benguela Current
- Current
- Ekman Layer
- Equatorial Undercurrent
- Gulf Stream
- Indian Ocean
- Kuroshio Current
- Meridional Overturning Circulation
- Mixed Layer
- Modeling of Ocean Circulation
- Pacific Ocean
- Peruvian Current
- Salinity
- Seawater, Composition of
- Somali Current
- Southern Ocean
- Thermocline
- Thermohaline Circulation
- Upwelling, Coastal
- Upwelling, Equatorial
- Western Boundary Currents
- Wind-Driven Circulation
- Paleoclimates
- Cenozoic Era
- Cretaceous Era
- Earth's Climate History
- Greenland Cores
- Holocene Era
- Jurassic Era
- Mesozoic Era
- Milankovitch Cycles
- Orbital Parameters, Eccentricity
- Orbital Parameters, Obliquity
- Orbital Parameters, Precession
- Paleozoic Era
- Pleistocene Era
- Pliocene Era
- Precambrian Era
- Quaternary Era
- Tertiary Climate
- Triassic Period
- Vostok Core
- Younger Dryas
- People
- Arakawa, Akio
- Arrhenius, Svante August
- Bolin, Bert
- Broecker, Wallace
- Bryan, Kirk
- Bryson, Reid
- Budyko, Mikhail
- Chamberlin, Thomas C.
- Charney, Jule Gregory
- Croll, James
- Fourier, Joseph
- Gore, Albert, Jr.
- Hadley, George
- Hansen, James
- Keeling, Charles David
- Lindzen, Richard
- Lorenz, Edward
- Manabe, Syukuro
- Milankovitch, Milutin
- Munk, Walter
- Phillips, Norman
- Revelle, Roger
- Richardson, Lewis Fry
- Rossby, Carl-Gustav
- Schneider, Stephen H.
- Singer, S. Fred
- Smagorinsky, Joseph
- Stommel, Henry
- Sverdrup, Harald Ulrik
- Tyndall, John
- Von Neumann, John
- Walker, Gilbert
- Washington, Warren
- Programs and Conventions
- Loading...
Get a 30 day FREE TRIAL
-
Watch videos from a variety of sources bringing classroom topics to life
-
Read modern, diverse business cases
-
Explore hundreds of books and reference titles

Sage Recommends
We found other relevant content for you on other Sage platforms.

Have you created a personal profile? Login or create a profile so that you can save clips, playlists and searches