Entry
Reader's guide
Entries A-Z
Subject index
Acid Rain
The critical issue of acid rain is a good example of a transnational environmental problem. An increase in acidic gases and particles in the atmosphere produced in one country can impact other countries further downwind of these acidifying emissions.
The acidic nature of rain was first observed near industrial cities in England and Scotland by an English chemist, Robert Angus Smith, in 1832. However, it was not until the late 1960s and early 1970s that acid rain was recognized as a widespread environmental problem in Europe and eastern North America. According to Gene Likens, F. Herbert Bormann, and Noye Johnson (1972), acid rain is a general or popular term, now known to include not only wet deposition from rain, snow, sleet, hail, cloud, and fog water but also dry deposition from acidifying particles and gases. Acid deposition is the more scientific term for this phenomenon.
The sources of most acid deposition are anthropogenic, mainly the burning of fossil fuels for power generation, industrial activity and transportation, and the smelting of metal ores. These activities produce SO2 and/or NOx (NO + NO2) gases, which are oxidized and converted to sulfuric and nitric acid, respectively. In their simplest forms, the reactions are:
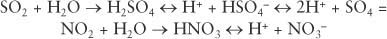
pH is a measure of hydrogen ion (H+) concentration, and the scale is logarithmic (pH = −log H+ concentration), so precipitation with a pH of 4.0 has 10 times more H+ ions (100 μeq/l) than precipitation with a pH of 5.0 (10 μeq/l). The pH of rain in industrialized regions is mainly the result of sulfuric and nitric acid. Precipitation is naturally somewhat acidic because of the carbonic acid from CO2 and organic acids from biological activity. Volcanic activity can increase atmospheric acidity by producing hydrochloric acid (HCl), as well as sulfuric acid. Lightning and forest fires can convert molecular nitrogen (N2), the main component of the atmosphere, to nitrogen oxides, which then can form nitric acid. These natural sources of acidity are generally small and reduce the pH of precipitation from 5.6 or higher to about 5.2. However, in many industrialized areas of the world, the average pH of precipitation is 4.0 to 4.5, with individual events often much lower. Organic acids can contribute to acidity (Likens, Keene, Miller, & Galloway, 1987), but their contribution is usually less than 20% of the total acidity in industrial areas.
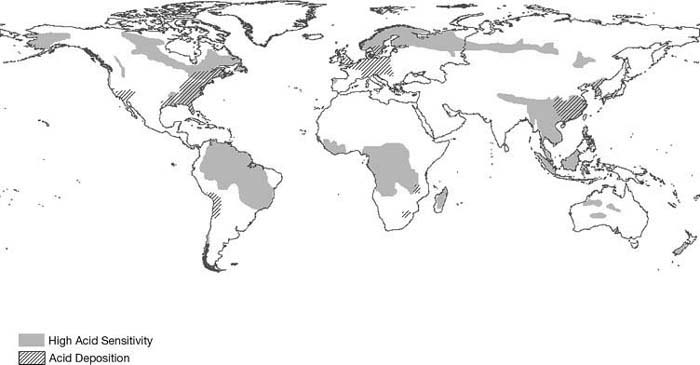
The largest amounts of acid deposition occur in eastern North America, central and northwestern Europe, and more recently in Southeast Asia (see Figure 1). These are all areas downwind of large emissions of SO2 and NOx resulting mainly from fossil fuel–based power production and industrial activities. China is the leading producer of SO2, having produced 33.2 million metric tons in 2006 (Lu et al., 2010), and the United States produces the most NOx, 16.6 million metric tons in 2005 (U.S. Environmental Protection Agency, 2010).
Figure 1 Areas sensitive to acid deposition (shaded) and areas receiving acid deposition (hatched areas show pH of rain < 4.5).;
...
- Activism, Transnational
- Amnesty International
- Anti-Apartheid Movement
- Antiglobalization Movements and Critics
- Antislavery Movements
- Associations
- Charities, Charity
- Civil Society, Global
- Civility
- Connectedness, Global
- Elites
- Foundations
- Foundations, Asian: International Activities
- Foundations, European: International Activities
- Foundations, U.S.: International Activities
- Gay and Lesbian Movement
- Humanitarian Intervention
- Humanitarianism
- Humanity, Concepts of
- Indigenous Peoples' Rights
- International Nongovernment Organizations (INGOs)
- International Nongovernment Organizations, Quasi-Forms
- Open Society Institute
- Opinion, World
- Philanthropy
- Publics and Polis, Global
- Red Cross
- Social Capital
- Social Entrepreneurship
- Social Movements
- Social Networking
- Solidarity Movements
- State–Civil Society Relations
- Uncivil Society
- UN–Civil Society Relations
- Waqfs
- Women's Movement
- World Economic Forum
- World Social Forum
- Air Travel
- Airlines
- Civil Aviation
- Communicative Power
- Computing
- Computing, Personal
- Containerization
- Cybernetics
- Global Communications and Technology
- Handheld Devices
- Information Age
- International Air Transportation Association
- Internet
- Internet Corporation for Assigned Names and Numbers
- Inventions and Discoveries
- Media, Global
- Morse Code
- Nanotechnology
- Navigation Systems
- Postal System, World
- Radio
- Railroads
- Road Signage Systems
- Roads and Routeways
- Silk Road, New
- Space, Outer
- Telegraph, Telegram
- Telephone
- Telephony
- Television
- Transportation Systems
- Universal Postal Union
- Web 2.0
- American Revolution
- Apartheid
- Cold War
- Conflict and Conflict Resolution
- Conquests
- Cooperation
- Cyberconflict
- Deterrence
- Failed States
- French Revolution
- Geneva Conventions
- Genocides
- Global Conflict and Security
- Hiroshima
- Holocaust
- Intelligence Agencies
- Military
- Pariahs, Global
- Peace
- Peace Activism
- Peacekeeping Forces
- Private Security Firms
- Refugees
- Religious Politics
- Revolutions
- Security
- September 11, 2001 (9/11)
- Soft Power Diplomacy
- Terrorism
- Treaties
- War
- War, Civil
- War, Urban
- Wars, New
- Wars, World
- Weapons
- Academy Awards
- Aesthetics
- Al Jazeera
- Americanization
- Architecture
- Art
- Artists
- Blogs
- British Broadcasting Corporation (BBC)
- Cable News Network (CNN)
- Cartoons, Comix, Manga
- Cinema
- Creativity and Innovation
- Creolization
- Cuisine
- Cultural Commons
- Cultural Destruction
- Cultural Diversity, Convention on
- Cultural Hybridity
- Cultural Industries
- Cultural Observatories
- Culture, Notions of
- Educational Systems
- Events, Global
- Film Festivals
- Games
- Genius, Notion of
- Geographic Informational Systems (GIS)
- Global Culture, Media
- Global Intellectuals
- Global Positioning System (GPS)
- Hajj
- Heritage
- Hollywood
- Homogenization
- Journalism
- Knowledge Management Systems
- Knowledge Production Systems
- Leisure
- Lifestyles
- Literature
- McDonaldization, McWorld
- Maps and Map-Making
- Memory
- Memory Wars
- Music
- Myths
- News
- Nobel Prize
- Olympic Movement
- Postmodernity
- Prizes and Awards, International
- Scripts and Writing Systems
- Sites, Global
- Soccer
- Sports, Recreation
- Standards and Standard Setting, Global
- Theater
- Think Tanks
- United Nations Educational, Scientific and Cultural Organization (UNESCO)
- Universities and Higher Learning
- Virtual Worlds
- Wikipedia
- Work
- World Cultures
- World's Fairs
- Acculturation
- Aging Societies
- Assimilation
- Baby Boomers
- Beirut
- Cities
- Communities, Transnational
- Contraception
- Demographic Change
- Demographic Transition
- Deterritorialization and Reterritorialization
- Diasporas
- Dubai
- Family Policies
- Family Systems, Kinship
- Fertility
- Hong Kong
- Immigration
- Immigration and Transnationalism
- Intergenerational Relations
- Johannesburg
- Landless Persons
- London
- Los Angeles
- Malthusian Idea
- Migrant Shopping Patterns
- Migration
- Migration, Illegal
- Migration Policies, Types of
- Mortality
- Mumbai
- New York City
- Overpopulation
- Paris
- Population and Demographic Change
- Population Control Policies
- Population Growth and Population Explosion
- Retirement Systems
- Rio de Janeiro
- Rurality
- Shanghai
- Shelter and Housing
- Singapore
- 68 Generation
- Tokyo
- Undocumented Persons
- Urban Diseconomies
- Urbanization
- Accounting Systems
- Agriculture Sector
- Asian Tiger Phenomenon
- Banking, Offshore
- Banks
- Capitalism
- Consumer Protest
- Consumerism
- Corporations, Transnational
- Currencies
- Data Systems and Reporting, Global
- Dependency
- Dependency Theory
- Depression, Great
- Depression, Recession, and Stagnation
- Deskilling
- Distribution of Wealth, Equitability of
- Dollar
- Economic Crises
- Economic Development
- Economic Ethics
- Economics, Keynesian
- Economy, Informal
- Entrepreneurship
- Euro
- European Central Bank
- Extractive Industries Sector
- Finance, Financial Systems
- Fordism
- Franchise Systems
- Global Economic Issues
- Globalization, Managed (China)
- Gold Standard
- Hedge Funds
- Illegal Trade, Arms
- Illegal Trade, Children
- Illegal Trade, Drugs
- Illegal Trade, Precious Metals
- Industrialization
- Inequality, Global
- Inequality, Global Economic
- International Labour Organization (ILO)
- International Monetary Fund (IMF)
- Investments
- Labor
- Local Exchange and Trading Systems (LETS)
- Longue Durrée, Long Wave Theories of Development
- Manufacturing Sector
- Maquiladoras
- Marketing
- Markets
- Marshall Plan
- McDonaldization, McWorld
- Mercantilism
- Microsoft
- Monetary Policy
- Organisation for Economic Co-operation and Development (OECD)
- Outsourcing
- Peasant Economies
- Petroleum Geopolitics
- Petroleum Economy
- Pharmaceutical Sector
- Poverty and Poverty Alleviation
- Private-Public Partnerships
- Privatization
- Production and Innovation Networks, Global
- Professions
- Protectionism
- Public Goods, Global
- Remittances
- Service Sector
- Sex Trafficking
- Slavery
- Tax Havens
- Taxation
- Technology Sector
- Tobin Tax
- Tourism Sector
- Trade
- Trade Agreements
- Value/Commodity Chains, Global
- Walmart
- World Bank
- World Trade Organization (WTO)
- Acid Rain
- Alternative Energy Sources
- Biofuels
- Biohazards
- Biological Diversity
- Biosphere
- Climate Change
- Deforestation
- Desertification
- Earth Summit
- Electricity
- Energy Efficiency
- Environmental Carrying Capacity
- Environmental Change
- Environmental Movement
- Environmental Rights
- Environmental Security
- Environmental Treaties, Conventions, and Protocols
- Extractive Industries Transparency Initiative
- Fisheries
- Floods, Tsunamis
- Forests
- Global Commons
- Global Environmental and Energy Issues
- Global Warming
- Greenhouse Gases
- International Maritime Organization
- Kyoto Protocol
- Land Use
- Natural Gas
- Nature, Concepts of
- Nuclear Power
- Oceans
- Oil
- Organization of Petroleum Exporting Countries (OPEC)
- Parks, Reserves, and Refuges
- Petroleum Geopolitics
- Polar Regions
- Remediation
- Sustainability
- United Nations Environment Programme (UNEP)
- Waste Management
- Water
- Accountability
- Arab League
- Association of Southeast Asian Nations (ASEAN)
- Borders
- Bretton Woods Agreements/System
- Caliphate
- Charismatic Leaders
- Civilization
- Colonialism
- Democracy
- Dynasties
- Empires
- Empires, Modern
- European Union
- Failed States
- Global Governance and World Order
- Global Order
- Global South
- Governance Networks, Transnational
- Hegemonic Power
- Hollow State
- Independence Movements
- International Relations
- Leadership
- League of Nations
- Legitimacy
- Methodological Nationalism
- Nation-State
- Neocolonialism
- Non-Aligned Movement
- North Atlantic Treaty Organization (NATO)
- Organization of American States (OAS)
- Pan African Union
- Petroleum Geopolitics
- Political Parties, Transnational
- Power, Global Contexts of
- Regional Governance
- Regionalism
- Republics
- Social Democracy
- Sovereignty
- Subsidiarity
- Summits, Summitry
- Transparency
- Treaty of Rome
- United Nations
- Utopia, Dystopia
- Vatican
- Welfare State
- Westphalia, Treaty of, and the Post-Westphalian World
- World Federalist Movement
- World Government
- World Order, Visions of
- Abortion
- Birth Control
- Burial and Crematory Practices
- Diseases
- Drugs and Pharmaceuticals
- Food
- Food and Agriculture Organization of the United Nations
- Global Health and Nutrition
- Health Care Access
- Health Care Systems
- HIV/AIDS
- Hygiene
- Infant Mortality
- Malnutrition
- Medical Systems
- Public Health
- Sexually Transmitted Diseases
- Tuberculosis
- Viruses, Killer
- Viruses and Diseases, Emerging
- World Food Program
- World Health Organization
- Global Historical Antecedents
- Global History, Studies in
- Animal Rights
- Arbitration
- Asylum
- Citizenship
- Civil Rights
- Constitutionalism
- Crime, Transnational
- Dharma
- Free Speech
- Gay Rights
- Global Justice and Legal Issues
- Human Rights, International
- Indigenous Peoples' Rights
- Intellectual Property Rights
- International Court of Justice
- International Criminal Tribunals
- Interpol
- Justice, Transitional
- Justice Movements, Transnational
- Labor Rights
- Law, International
- Law, Regional
- Law, Transnational
- Law, World
- Law of Arctic Regions
- Law of the Sea
- Legal Systems
- Lex Mercatoria
- Natural Law
- Nuremburg Precedent
- Penal Systems
- Policing Systems
- Prisoners' Rights
- Shari'a (Islamic Law)
- Truth Commission
- Universal Jurisdiction
- Women's Rights
- African Diaspora Religions
- African Religions
- Baha'i
- Battle of Badr
- Buddhism
- Capitalism
- Christianity
- Christianity-Related Movements
- Communism, as International Movement
- Communist International
- Communitarianism
- Confucianism
- Cosmopolitanism
- Crusades
- Darwinism and Social Darwinism
- Enlightenment, The
- Ethics, Global
- Fascism
- Feminism
- Freemasons
- Global Religions, Beliefs, Ideologies
- Hare Krishna (International Society for Krishna Consciousness)
- Hinduism
- Hindu-Related Movements
- Humanism
- Idealism
- Ideologies, Global
- Imperialism
- Indigenous Religions, Globalization of
- Individualism
- Islam
- Islam-Related Movements
- Jainism
- Judaism
- Liberalism, Neoliberalism
- Marxism and Neo-Marxism
- Modernization
- Mormonism
- Myths
- Nationalism, Neo-Nationalism
- Neoconservatism
- Populism
- Postmodernism
- Protestant Reformation
- Religious Conversion
- Religious Movements, New and Syncretic
- Secularism
- Shinto
- Sikhism
- Socialism
- Socialist International
- Third Way Movements
- World Religions, Concept of
- Zionism
- Zoroastrianism
- Civil Society, Global
- Demographic Change
- Global Communications and Technology
- Global Conflict and Security
- Global Culture, Media
- Global Economic Issues
- Global Environmental and Energy Issues
- Global Governance and World Order
- Global Health and Nutrition
- Global Historical Antecedents
- Global History, Studies in
- Global Justice and Legal Issues
- Global Order
- Global Religions, Beliefs, and Ideologies
- Global Reporting Initiatives
- Global Studies
- Global Studies, Current Academic Approaches to
- Global Studies, Early Academic Approaches to
- Global Terminology
- Globalization, Approaches to
- Globalization, Measurement of
- Globalization, Phenomenon of
- Globalization and Transnationality Indexes
- Hyperglobalism
- Identities in Global Society
- Inequality, Global
- Proto-Globalization
- Shrinking World Concepts
- World Order, Visions of
- World Society Theory
- World-Systems Perspective
- Class
- Community
- Corporate Identity
- Cosmopolitan Identity
- Ethnic Identity
- Ethnocentrism
- Family
- Family Systems, Kinship
- Gender Identity
- Global Village
- Homophobia
- Identities, Traditional
- Identities in Global Society
- Linguistic Identities
- Marginality
- Modern Identities
- Multiculturalism
- Multiracial Identities
- National Identities
- Otherness
- Racial Identity
- Racial Supremacy
- Regional Identities
- Religious Identities
- Secret Societies
- Tribal Identities
- Universalism
- Values
- Xenophobia
- Loading...
Get a 30 day FREE TRIAL
-
Watch videos from a variety of sources bringing classroom topics to life
-
Read modern, diverse business cases
-
Explore hundreds of books and reference titles

Sage Recommends
We found other relevant content for you on other Sage platforms.

Have you created a personal profile? Login or create a profile so that you can save clips, playlists and searches