Entry
Reader's guide
Entries A-Z
Subject index
Case-Cohort Studies
The case-cohort design was proposed by Ross Prentice as an alternative design in epidemiologic follow-up studies that is less expensive than a full-scale cohort study. The case-cohort design involves collecting covariate data only for cases, that is, subjects who experience the event of interest in a cohort, and for members of a relatively small randomly selected subcohort. The subcohort may serve as a comparison group for several different types of disease outcomes.
The case-cohort design can substantially reduce cost and effort of exposure assessment by limiting exposure to a small fraction of the cohort with only a small loss of efficiency compared with a full cohort design. The case-cohort design is most beneficial when the most expensive part of the study is not in ascertaining subjects but in measuring their exposures. If the main cost is in ascertainment, a full cohort analysis might be a more sensible approach to analysis.
The design is particularly suited to settings such as molecular epidemiologic studies, where raw materials for covariate information, for example, biospecimens, can be collected and stored. For the cases, these specimens can then be analyzed after the failure, that is, event of interest, has occurred, to determine what an individual's levels of exposure were at the times before failure. The case-cohort design has been applied in cancer research, cardiovascular disease, and HIV research and has become increasingly popular in genetic epidemiologic studies.
Most methods used to study relative risks in failure time models are based on the Cox proportional hazards model, which assumes a multiplicative form for the model of disease occurrence
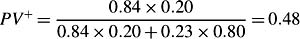
where x(t) denotes covariates at time t, λ0(t)standsfor the baseline hazard rate for subjects with x = 0, and rr denotes the relative risk part, with rr(Xi(t), 0) = 1. Under the case-cohort design, most existing relative risk estimators are based on modifications of the full cohort partial likelihood score functions, by weighing the contributions from cases and subcohort members by the inverse of their true or estimated sampling probabilities. In the pseudo-likelihood approach proposed by Ross Prentice, for each failure, a sampled risk set is formed by the case and the controls who are in the subcohort. Subcohort members contribute to the analysis over the entire time on study, but the failures outside the subcohort contribute only at their failure times. The pseudo-likelihood contribution is based on the conditional probability that the case fails given that someone fails among those sampled into the risk set. The pseudo likelihood is then the product of the conditional probabilities over failure times. Letting Yi denote the ‘at-risk’ indicator of the ith subject at the failure time and rr the relative risk part, the pseudo likelihood is given by
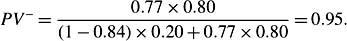
This pseudo likelihood differs from the partial likelihood for a full cohort study in that the denominator is summing over subjects at risk in the subcohort rather than subjects at risk in the entire cohort. In addition, as cases are added at the time of event, the risk sets are not nested.
The score of the pseudo likelihood has expected value of 0 at the true value of b, but the inverse information does not estimate the variance of the estimator due to sampling-induced covariances between the score terms. The estimator has an asymptotic normal distribution and several approaches to variance estimation are available. Standard Cox regression software can be used to estimate parameters for case-cohort samples; however, variance computations need to be adapted to accommodate the design, which can be done using the delta beta option available in many statistics software packages.
...
- Behavioral and Social Science
- Acculturation
- Bioterrorism
- Community Health
- Community Trial
- Community-Based Participatory Research
- Cultural Sensitivity
- Demography
- Determinants of Health Model
- Ecological Fallacy
- Epidemiology in Developing Countries
- EuroQoL EQ-5D Questionnaire
- Functional Status
- Genocide
- Geographical and Social Influences on Health
- Health Behavior
- Health Belief Model
- Health Communication
- Health Communication in Developing Countries
- Health Disparities
- Health Literacy
- Health, Definitions of
- Life Course Approach
- Locus of Control
- Medical Anthropology
- Network Analysis
- Participatory Action Research
- Poverty and Health
- Quality of Life, Quantification of
- Quality of Well-Being Scale (QWB)
- Race and Ethnicity, Measurement Issues With
- Race Bridging
- Rural Health Issues
- Self-Efficacy
- SF-36® Health Survey
- Social Capital and Health
- Social Epidemiology
- Social Hierarchy and Health
- Social Marketing
- Social-Cognitive Theory
- Socioeconomic Classification
- Spirituality and Health
- Targeting and Tailoring
- Theory of Planned Behavior
- Transtheoretical Model
- Urban Health Issues
- Urban Sprawl
- Branches of Epidemiology
- Applied Epidemiology
- Chronic Disease Epidemiology
- Clinical Epidemiology
- Descriptive and Analytic Epidemiology
- Disability Epidemiology
- Disaster Epidemiology
- Eco-Epidemiology
- Environmental and Occupational Epidemiology
- Field Epidemiology
- Genetic Epidemiology
- Injury Epidemiology
- Maternal and Child Health Epidemiology
- Molecular Epidemiology
- Neuroepidemiology
- Nutritional Epidemiology
- Pharmacoepidemiology
- Psychiatric Epidemiology
- Reproductive Epidemiology
- Social Epidemiology
- Veterinary Epidemiology
- Diseases and Conditions
- Alzheimer's Disease
- Anxiety Disorders
- Arthritis
- Asthma
- Autism
- Avian Flu
- Bipolar Disorder
- Bloodborne Diseases
- Cancer
- Cardiovascular Disease
- Diabetes
- Foodborne Diseases
- Gulf War Syndrome
- Hepatitis
- HIV/AIDS
- Hypertension
- Influenza
- Insect-Borne Disease
- Malaria
- Measles
- Oral Health
- Osteoporosis
- Parasitic Diseases
- Plague
- Polio
- Post-Traumatic Stress Disorder
- Schizophrenia
- Severe Acute Respiratory Syndrome (SARS)
- Sexually Transmitted Diseases
- Sick Building Syndrome
- Sleep Disorders
- Smallpox
- Suicide
- Toxic Shock Syndrome
- Tuberculosis
- Vector-Borne Disease
- Vehicle-Related Injuries
- Vitamin Deficiency Diseases
- Waterborne Diseases
- Yellow Fever
- Zoonotic Disease
- Epidemiological Concepts
- Attack Rate
- Attributable Fractions
- Biomarkers
- Birth Cohort Analysis
- Birth Defects
- Case Definition
- Case Reports and Case Series
- Case-Cohort Studies
- Case-Fatality Rate
- Cohort Effects
- Community Trial
- Competencies in Applied Epidemiology for Public Health Agencies
- Cumulative Incidence
- Direct Standardization
- Disease Eradication
- Effect Modification and Interaction
- Effectiveness
- Efficacy
- Emerging Infections
- Epidemic
- Etiology of Disease
- Exposure Assessment
- Fertility, Measures of
- Fetal Death, Measures of
- Gestational Age
- Health, Definitions of
- Herd Immunity
- Hill's Considerations for Causal Inference
- Incidence
- Indirect Standardization
- Koch's Postulates
- Life Course Approach
- Life Expectancy
- Life Tables
- Malnutrition, Measurement of
- Mediating Variable
- Migrant Studies
- Mortality Rates
- Natural Experiment
- Notifiable Disease
- Outbreak Investigation
- Population Pyramid
- Preclinical Phase of Disease
- Preterm Birth
- Prevalence
- Prevention: Primary, Secondary, and Tertiary
- Public Health Surveillance
- Qualitative Methods in Epidemiology
- Quarantine and Isolation
- Screening
- Sensitivity and Specificity
- Sentinel Health Event
- Syndemics
- Epidemiologic Data
- Administrative Data
- American Cancer Society Cohort Studies
- Behavioral Risk Factor Surveillance System
- Biomedical Informatics
- Birth Certificate
- Cancer Registries
- Death Certificate
- Framingham Heart Study
- Global Burden of Disease Project
- Harvard Six Cities Study
- Health Plan Employer Data and Information Set
- Healthcare Cost and Utilization Project
- Healthy People 2010
- Honolulu Heart Program
- Illicit Drug Use, Acquiring Information on
- Medical Expenditure Panel Survey
- Monitoring the Future Survey
- National Ambulatory Medical Care Survey
- National Death Index
- National Health and Nutrition Examination Survey
- National Health Care Survey
- National Health Interview Survey
- National Immunization Survey
- National Maternal and Infant Health Survey
- National Mortality Followback Survey
- National Survey of Family Growth
- Physicians' Health Study
- Pregnancy Risk Assessment and Monitoring System
- Relational Database
- Rochester Epidemiology Project
- Sampling Techniques
- Secondary Data
- Spreadsheet
- Youth Risk Behavior Surveillance System
- Ethics
- Genetics
- Association, Genetic
- Chromosome
- Epigenetics
- Family Studies in Genetics
- Gene
- Gene-Environment Interaction
- Genetic Counseling
- Genetic Disorders
- Genetic Epidemiology
- Genetic Markers
- Genomics
- Genotype
- Hardy-Weinberg Law
- Heritability
- Human Genome Project
- Icelandic Genetics Database
- Linkage Analysis
- Molecular Epidemiology
- Multifactorial Inheritance
- Mutation
- Newborn Screening Programs
- Phenotype
- Teratogen
- Twin Studies
- Health Care Economics and Management
- Biomedical Informatics
- EuroQoL EQ-5D Questionnaire
- Evidence-Based Medicine
- Formulary, Drug
- Functional Status
- Health Care Delivery
- Health Care Services Utilization
- Health Economics
- International Classification of Diseases
- International Classification of Functioning, Disability, and Health
- Managed Care
- Medicaid
- Medicare
- Partner Notification
- Quality of Life, Quantification of
- Quality of Well-Being Scale (QWB)
- SF-36® Health Survey
- Health Risks and Health Behaviors
- Agent Orange
- Alcohol Use
- Allergen
- Asbestos
- Bioterrorism
- Child Abuse
- Cholesterol
- Circumcision, Male
- Diabetes
- Drug Abuse and Dependence, Epidemiology of
- Eating Disorders
- Emerging Infections
- Firearms
- Foodborne Diseases
- Harm Reduction
- Hormone Replacement Therapy
- Intimate Partner Violence
- Lead
- Love Canal
- Malnutrition, Measurement of
- Mercury
- Obesity
- Oral Contraceptives
- Pain
- Physical Activity and Health
- Pollution
- Poverty and Health
- Radiation
- Sexual Risk Behavior
- Sick Building Syndrome
- Social Capital and Health
- Social Hierarchy and Health
- Socioeconomic Classification
- Spirituality and Health
- Stress
- Teratogen
- Thalidomide
- Tobacco
- Urban Health Issues
- Urban Sprawl
- Vehicle-Related Injuries
- Violence as a Public Health Issue
- Vitamin Deficiency Diseases
- War
- Waterborne Diseases
- Zoonotic Disease
- History and Biography
- Budd, William
- Doll, Richard
- Ehrlich, Paul
- Epidemiology, History of
- Eugenics
- Farr, William
- Frost, Wade Hampton
- Genocide
- Goldberger, Joseph
- Graunt, John
- Hamilton, Alice
- Hill, Austin Bradford
- Jenner, Edward
- Keys, Ancel
- Koch, Robert
- Lind, James
- Lister, Joseph
- Nightingale, Florence
- Pasteur, Louis
- Public Health, History of
- Reed, Walter
- Ricketts, Howard
- Rush, Benjamin
- Snow, John
- Tukey, John
- Tuskegee Study
- Infrastructure of Epidemiology and Public Health
- American College of Epidemiology
- American Public Health Association
- Association of Schools of Public Health
- Centers for Disease Control and Prevention
- Council of State and Territorial Epidemiologists
- European Public Health Alliance
- European Union Public Health Programs
- Food and Drug Administration
- Governmental Role in Public Health
- Healthy People 2010
- Institutional Review Board
- Journals, Epidemiological
- Journals, Public Health
- National Center for Health Statistics
- National Institutes of Health
- Pan American Health Organization
- Peer Review Process
- Public Health Agency of Canada
- Publication Bias
- Society for Epidemiologic Research
- Surgeon General, U.S.
- U.S. Public Health Service
- United Nations Children's Fund
- World Health Organization
- Medical Care and Research
- Allergen
- Apgar Score
- Barker Hypothesis
- Birth Defects
- Body Mass Index (BMI)
- Carcinogen
- Case Reports and Case Series
- Clinical Epidemiology
- Clinical Trials
- Community Health
- Community Trial
- Comorbidity
- Complementary and Alternative Medicine
- Effectiveness
- Efficacy
- Emerging Infections
- Etiology of Disease
- Evidence-Based Medicine
- Gestational Age
- Intent-to-Treat Analysis
- International Classification of Diseases
- International Classification of Functioning, Disability, and Health
- Latency and Incubation Periods
- Life Course Approach
- Malnutrition, Measurement of
- Medical Anthropology
- Organ Donation
- Pain
- Placebo Effect
- Preclinical Phase of Disease
- Preterm Birth
- Public Health Nursing
- Quarantine and Isolation
- Screening
- Vaccination
- Specific Populations
- African American Health Issues
- Aging, Epidemiology of
- American Indian Health Issues
- Asian American/Pacific Islander Health Issues
- Breastfeeding
- Child and Adolescent Health
- Epidemiology in Developing Countries
- Hormone Replacement Therapy
- Immigrant and Refugee Health Issues
- Latino Health Issues
- Maternal and Child Health Epidemiology
- Men's Health Issues
- Oral Contraceptives
- Race and Ethnicity, Measurement Issues With
- Race Bridging
- Rural Health Issues
- Sexual Minorities, Health Issues of
- Urban Health Issues
- Women's Health Issues
- Statistics and Research Methods
- F Test
- p Value
- Additive and Multiplicative Models
- Analysis of Covariance
- Analysis of Variance
- Bar Chart
- Bayes's Theorem
- Bayesian Approach to Statistics
- Bias
- Binomial Variable
- Birth Cohort Analysis
- Box-and-Whisker Plot
- Capture-Recapture Method
- Categorical Data, Analysis of
- Causal Diagrams
- Causation and Causal Inference
- Censored Data
- Central Limit Theorem
- Chi-Square Test
- Classification and Regression Tree Models
- Cluster Analysis
- Coefficient of Determination
- Cohort Effects
- Collinearity
- Community Trial
- Community-Based Participatory Research
- Confidence Interval
- Confounding
- Control Group
- Control Variable
- Convenience Sample
- Cox Model
- Critical Value
- Cumulative Incidence
- Data Management
- Data Transformations
- Decision Analysis
- Degrees of Freedom
- Dependent and Independent Variables
- Diffusion of Innovations
- Discriminant Analysis
- Dose-Response Relationship
- Doubling Time
- Dummy Coding
- Dummy Variable
- Ecological Fallacy
- Economic Evaluation
- Effect Modification and Interaction
- Factor Analysis
- Fisher's Exact Test
- Geographical and Spatial Analysis
- Graphical Presentation of Data
- Halo Effect
- Hawthorne Effect
- Hazard Rate
- Healthy Worker Effect
- Hill's Considerations for Causal Inference
- Histogram
- Hypothesis Testing
- Inferential and Descriptive Statistics
- Intent-to-Treat Analysis
- Internet Data Collection
- Interquartile Range
- Interrater Reliability
- Intervention Studies
- Interview Techniques
- Kaplan-Meier Method
- Kappa
- Kurtosis
- Latent Class Models
- Life Tables
- Likelihood Ratio
- Likert Scale
- Log-Rank Test
- Logistic Regression
- Longitudinal Research Design
- Matching
- Measurement
- Measures of Association
- Measures of Central Tendency
- Measures of Variability
- Meta-Analysis
- Missing Data Methods
- Multilevel Modeling
- Multiple Comparison Procedures
- Multivariate Analysis of Variance
- Natural Experiment
- Network Analysis
- Nonparametric Statistics
- Normal Distribution
- Null and Alternative Hypotheses
- Observational Studies
- Overmatching
- Panel Data
- Participatory Action Research
- Pearson Correlation Coefficient
- Percentiles
- Person-Time Units
- Pie Chart
- Placebo Effect
- Point Estimate
- Probability Sample
- Program Evaluation
- Propensity Score
- Proportion
- Qualitative Methods in Epidemiology
- Quasi Experiments
- Questionnaire Design
- Race Bridging
- Random Variable
- Random-Digit Dialing
- Randomization
- Rate
- Ratio
- Receiver Operating Characteristic (ROC) Curve
- Regression
- Relational Database
- Reliability
- Response Rate
- Robust Statistics
- Sample Size Calculations and Statistical Power
- Sampling Distribution
- Sampling Techniques
- Scatterplot
- Secondary Data
- Sensitivity and Specificity
- Sequential Analysis
- Simpson's Paradox
- Skewness
- Spreadsheet
- Stem-and-Leaf Plot
- Stratified Methods
- Structural Equation Modeling
- Study Design
- Survival Analysis
- Target Population
- Time Series
- Type I and Type II Errors
- Unit of Analysis
- Validity
- Volunteer Effect
- Loading...
Get a 30 day FREE TRIAL
-
Watch videos from a variety of sources bringing classroom topics to life
-
Read modern, diverse business cases
-
Explore hundreds of books and reference titles

Sage Recommends
We found other relevant content for you on other Sage platforms.

Have you created a personal profile? Login or create a profile so that you can save clips, playlists and searches