Entry
Entries A-Z
Subject index
CALCIUM (CA)
The fifth most abundant element by mass in the Earth’s crust, the fifth most abundant dissolved ion in seawater and the most abundant metal in the human body. Calcium does not occur naturally in its metallic state and is found associated with other elements or in its ionic state. In minerals, calcium is sequestered in relatively insoluble compounds but is relatively mobile in its ionic state and calcium cations are found in freshwater and seawater. Calcium is found in sedimentary rocks as calcium carbonate and in igneous rocks as calcium silicates. It is essential for almost all forms of life and is the major element in the mineralisation of bones and shells as well as in plant cell wall development. In marine environments, calcium carbonate forms the skeletal material of most zooplankton and, in its crystalline form (aragonite), comprises coral. World production of the metal (used as an alloying agent) is only ~2,000 tonnes/year, whereas that of lime—calcium oxide, CaO—(used widely in cement manufacture, metallurgy, water treatment and the chemical industry) is ~120 million tonnes. Lime has been used since prehistoric times in mortar (a mixture of lime, sand and water, which hardens on exposure to the atmosphere as it converts to calcium carbonate by absorbing carbon dioxide) and in soils to neutralise acidity for agriculture.
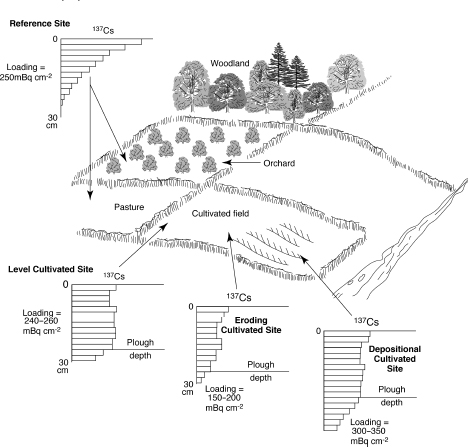
Caesium-137Schematic representation of the effects of various agricultural practices on the loading and distribution within the soil profile of caesium-137 (Walling and Quine, 1990).
[See alsocalcium cycle, limestone, liming, seawater composition, teeth]
- Loading...
Get a 30 day FREE TRIAL
-
Watch videos from a variety of sources bringing classroom topics to life
-
Read modern, diverse business cases
-
Explore hundreds of books and reference titles

Sage Recommends
We found other relevant content for you on other Sage platforms.

Have you created a personal profile? Login or create a profile so that you can save clips, playlists and searches