Entry
Entries A-Z
Subject index
ACID SOILS
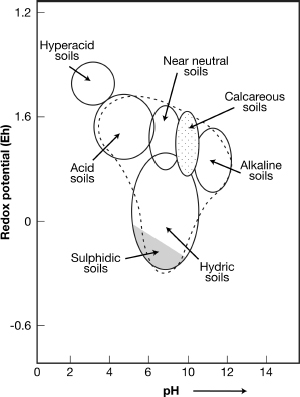
Soil acidity varies from about pH 2.0–10.0, acid soils have a pH of <7.0 but most acid soils with a wide distribution (acrisols, ferralsols and podzols) have a pH of 3.5–4.0 (see Figure). In the soil types with a wide distribution, the acidity comes from weakly acid rain water and is accentuated by the metabolism of roots and micro-organisms. In hyperacid soils the higher acidity levels come from the oxidation of sulfide-rich minerals, such as pyrites. High acidity is associated with low cation exchange capacity (CEC) and low base saturation, which lead to agricultural problems from the toxicity of aluminium (Al) and nutrient deficiency.
Acid soilsVarious types of soils in relation to their acidity (pH) and redox potential (Eh). The envelope for common mineral soils is indicated by the dashed line (as used in Chesworth, 2008).
[See alsoacid rain, acid sulfate soils, acidification, liming]
- Loading...
Get a 30 day FREE TRIAL
-
Watch videos from a variety of sources bringing classroom topics to life
-
Read modern, diverse business cases
-
Explore hundreds of books and reference titles

Sage Recommends
We found other relevant content for you on other Sage platforms.

Have you created a personal profile? Login or create a profile so that you can save clips, playlists and searches