Entry
Entries A-Z
Subject index
ACID RAIN
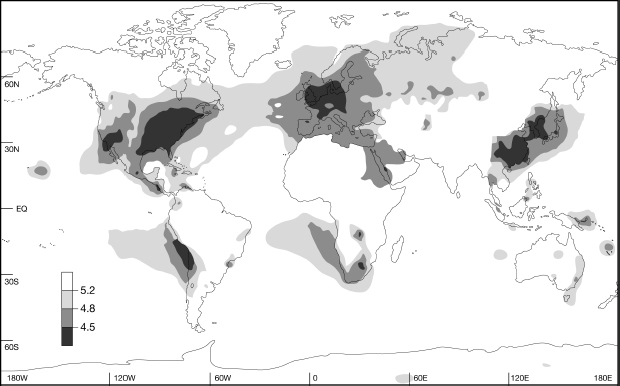
precipitation with an artificially low pH due to industrial emissions. The pH of pure water in equilibrium with atmospheric carbon dioxide is 5.6 and thus the term acid rain is reserved for precipitation of pH <5.6. There has been a notable increase in the acidity of precipitation across North America and Europe since the industrial revolution due to the emission of sulfur dioxide and nitrogen oxides from the burning of fossil fuels. New regions with significant acid rain problems are emerging with the spread of industrialisation (see Figure). The oxidation of sulfur and nitrogen oxides in the atmosphere to sulfuric acid (H2SO4) and nitric acid (HNO3) creates relatively strong acids. It is estimated that 60–70 per cent of the mean annual acidity of precipitation originates from sulfuric acid and the remainder from nitric acid. This reflects the much larger quantities of sulfur emitted and the fact that the sulfuric acid molecule in solution releases two H+ ions and the nitric acid molecule only one. Precipitation in Scotland and North America with a pH as low as 2.1–2.4 has been recorded. Acid rain leads to accelerated leaching of base cations from soils, increased solubility of metals and the acidification of surface waters.
Acid rainAnnual average pH of precipitation at the global scale (Noone, 2012).
[See alsoforest decline, wet deposition]
- Loading...
Get a 30 day FREE TRIAL
-
Watch videos from a variety of sources bringing classroom topics to life
-
Read modern, diverse business cases
-
Explore hundreds of books and reference titles

Sage Recommends
We found other relevant content for you on other Sage platforms.

Have you created a personal profile? Login or create a profile so that you can save clips, playlists and searches